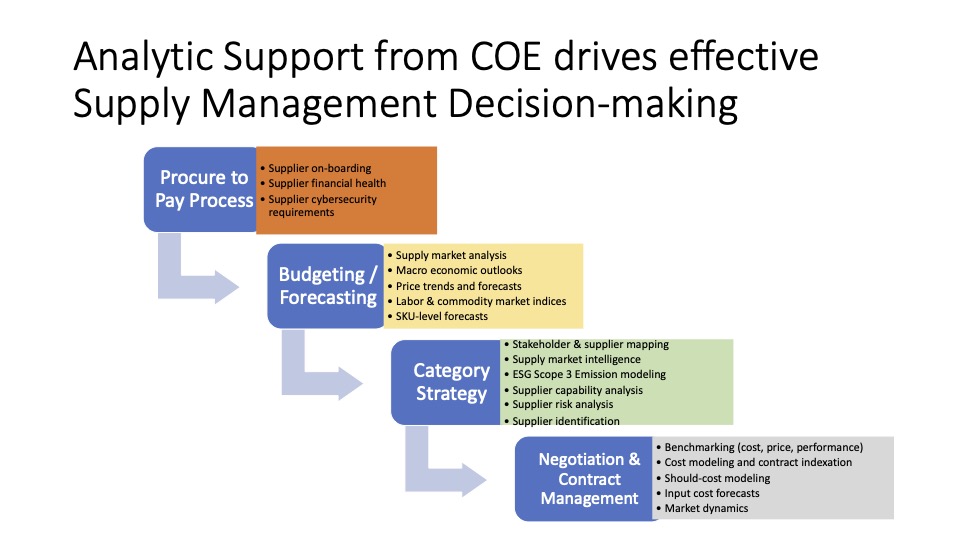
Just How Dire is the COVID Healthcare Global Supply Chain Response? (A Case Study)
One would surmise from some of the political proclamations made in the last few days that the COVID crisis is behind us and that everything is back to normal. Some of my recent research has involved interviews with healthcare physicians, distributors, manufacturers, and suppliers, and these discussions all point to a reality that is very different from what politicians would have us believe.
One executive we spoke with works for a distributor to different DoD and state agencies. They have been working to distribute PPE to these customers, and sought to begin distribution of test kits back in April. At that time, the FDA was issuing Emergency Use Authorization to several test kits. One company in California had developed a test kit that had 98% specificity, (99% compared to a PCR test). The distributor developed a partnership with this company to distribute to 50 states, subject to the EUA being issued. That was last April… and here we are in August, and the FDA has still not issued the EUA. This is because the FDA changed the guidelines after a host of companies had issued the tests for EUA’s in April. This change mandated that a third party testing body had to validate the tests, after everyone had already submitted their application for the EUA. HHS never provided guidance on which third party testing bodies could be used – and so the states were left once again to decide on their own what to do. They looked to their own departments of public health, who were also unsure about how to proceed.
One state, California, decided to go with the Optum Serve Federal Health Services to issue the tests, paying almost a billion dollars for 850,000 tests. This effort was incredibly unsuccessful. For instance, a drive through clinic in Los Angeles purchased 50,000 tests a week, but were only getting about 8,000 people to show up. The test location was off a freeway, and was not serviced by public transportation. (What Uber driver is going to take a person to a test site for a test?) One could only book a test between 9 and 4, with half hour appointments – and if you waited two weeks and were lucky you could get a test! The executive noted that “I made four phone calls to my insurance company, Kaiser, and was denied each time because I was too young, and finally convinced someone to let me get a test.”
This executives notes that “We serve eight states, and testing is pretty broken everywhere. There is no centralized entity that is buying tests for master distribution. Every entity is sort of on their own to procure a test they think is needed, and of course the model is even more complicated because the health systems must bow to the insurance companies, who will only pay for that test for which they have their own oversight, and which they will allow, as well as the fee and reimbursement for that test. Right now there are over 100 different PCR tests available, and no one knows which one is better than the others.”
The demand for PPE has also never let up, and the challenges of getting it have also never abated. As we’ve noted in prior blogs, almost all of the manufacturers are located in SE Asia. 3M has a 6 month waiting list to buy masks, and the cost of domestic masks is around $4-5. However, most of 3M’s masks are no longer produced by 3M, but are outsourced to Mexican companies like Moldex. 3M is also telling most hospitals there is a 9 month wait time. Switching to another domestic supplier like Dupont or Dow will take time, and who is willing to invest millions of dollars capital in mask production facilities, not knowing what the demand will be in the future?
This executive noted that “We are getting masks in the low $2 range delivered into the US. However the biggest need is nitrile gloves – as hospitals are using them, food service is using them, and everyone else as well. There are only a handful of factories that produce a total of 1 million boxes per month each, yet global demand for gloves is in the billions of boxes per month. If we want to order from Asia, they won’t take an order under 1 million boxes, which is 100 million gloves! The shipping time is 23 days at sea, and depending on customs clearance – the time from PO issued to possession is around 35 days. If you want to air freight a box, it is $3 a box, and you can only stuff 247,000 boxes in a 747. So for the minimum order, you need to put it on four planes, which amounts to $1.2 million for a single flight. Just try to get four flights back to back however, it is impossible. Every single flight is booked for weeks out. No one is going to begin producing nitrile gloves or gowns (that sell for $1.50) in the US, so it is unlikely we will be able to build domestic production around these items. We have also started to see some governments in southeast Asia begin to nationalize their glove production, as they are seeing demand at capacity for the next six months. As they nationalize, glove prices are going up to 5X of what they were previously. Also our government doesn’t issue payment before net 30. Manufacturers require 50% deposit to start the order, and 50% after it is produced, but before it’s left the country. So with 23 days by sea and net 30, you have to come up with capital for the gloves 60 days before you are paid, floating tens of millions of dollars. Financing this through third parties will of course increase the cost, and with the risks of nationalization, the financing fees are also going up…
Looking into the future, the next big issue is going to be vaccine distribution. One of the big bottlenecks is going to be syringes. We estimate the federal government will need 800 million syringes – which includes breakage, and the fact that everyone needs two shots. All syringe manufacture is in China, and the tariffs are still 7.5% on these items. So we anticipated this, and we started to look at India, which is the only country that can scale up on syringe manufacturing. But like masks, I anticipate there will be lots of speculation and third party brokers in the mix. Currently, the Strategic National Stockpile (SNS) contains about 15 million needles and syringes, about 2% of what will be needed, according to Bright’s complaint.
My takeaway from this discussion, is that we really need a strong centralized healthcare response. A vaccine that can’t be distributed properly is not going to solve the COVID crisis.